|
|

|
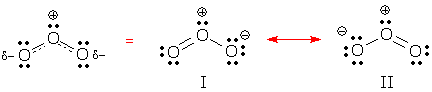
Localized vs. delocalized The distinction between "localized" and "delocalized" only exists when we describe electron motion using Lewis' bonding theory. According to this theory, localized electrons exhibit normal behavior. A localized lone pair remains close to one atom. A localized bond pair travels between two atoms. Resonance hybrids necessarily contain some "abnormal" electrons. A lone pair may seem to have some bonding characteristics; instead of sticking near one atom, it visits two atoms. A bond pair may appear to move between two different pairs of atoms. These electrons step outside the boundaries that Lewis' theory has set for them, and we consider them to be delocalized. The easiest way to spot delocalized electrons is to compare electron locations in two resonance forms. If a pair appears in one place in one form, and in a different place in another form, the pair is delocalized. You can see delocalized behavior in resonance forms I and II below. Both forms contain two delocalized electron pairs. Can you find them? What kind of electron pairs are delocalized in each structure?
(Answer: these forms contain one delocalized bond pair and one delocalized lone pair.)
Delocalized bonds and delocalized charges The "delocalized" label also applies to bonds and charges. A delocalized bond is a bond that appears in some resonance forms, but not others. Resonance form I contains 2 localized bonds and 1 delocalized bond. A delocalized charge is a formal charge that appears on one atom in some resonance forms and on other atoms in other forms. Ozone's negative charge is delocalized over the two end O, while the positive charge is localized on the center O. |