|
|

|
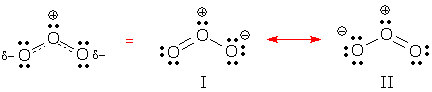
Resonance contributors or forms? A resonance hybrid can be drawn using a dashed-line formula, but these drawings make it hard to count electrons. Chemists prefer to draw a resonance hybrid as a superposition (or combination) of Lewis structures. The individual Lewis structures that appear in one of these superposition drawings are called resonance contributors or resonance forms. The following drawing of ozone illustrates both drawing methods. A dashed-line formula is shown on the left. A superposition of two resonance contributors (formulas I and II) is shown on the right:
A superposition drawing always includes at least two resonance contributors and a double-headed arrow connecting pairs of contributors. This arrow is a special symbol that means, "construct a superposition of the electron patterns connected by this arrow."
Deceptive Advertising Resonance forms have two flaws: they are deceptive and they ... are ... deceptive. Resonance forms, like normal Lewis structures, are always drawn using standard symbols. The drawing does not identify delocalized electrons or differentiate them from localized electrons. This is deceptive. Also, a resonance form necessarily presents a picture of the hybrid that is incomplete and incorrect. This is certainly deceptive. One way to generate the correct picture for a resonance hybrid is to draw a dashed-line formula. Alternatively, we can construct a superposition of the electron patterns shown in each resonance contributor. For example, we can find the charge on Oleft by averaging the neutral Oleft in I and the negative Oleft in II. This gives a partial negative charge. In the same way, we can tell that Oleft-Ocenter are really connected by a partial double bond because the bond is a superposition of the double bond in I and the single bond in II. Just remember this: even though resonance contributors look like Lewis structures, their status is different. A Lewis structure shows what a normal molecule looks like, but a resonance contributor does not. A resonance contributor is, by definition, a poor picture. You can think of a resonance contributor as being like a black-and-white photo. Useful, but misleading.
*Photo credit: http://www.georgeclooney.org/html/PhotoIndex.html
|